Transform Regulatory Complexity into
Strategic Advantage
RuleMedi is the agentic AI platform for Life Sciences. We turn the complexities of EU MDR, IVDR, and UKCA regulations into inspection-ready clarity and continuous assurance — across every stage of the device lifecycle.
Solutions
Compliance Pillars
Modes
Jurisdictions
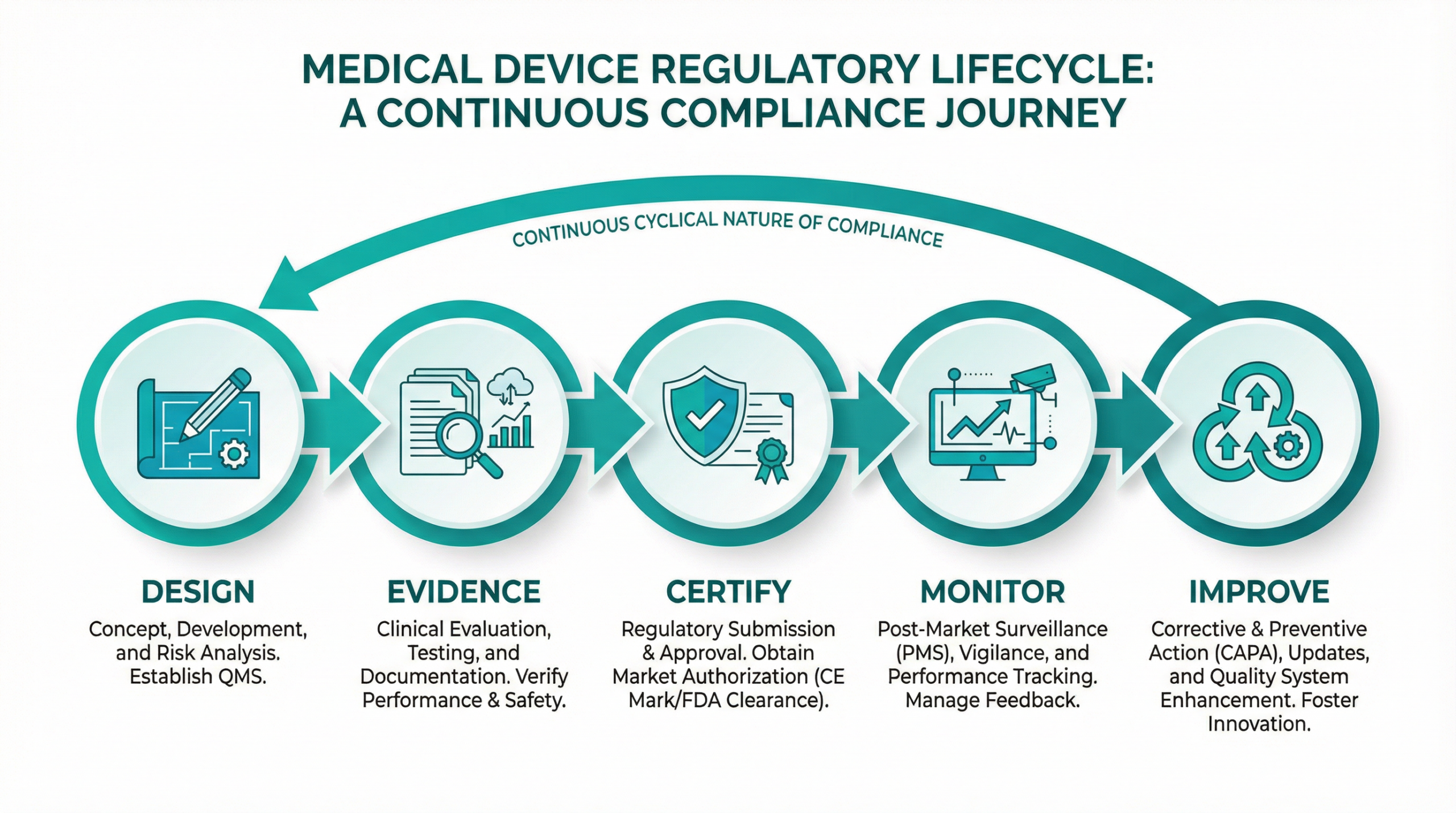
From Reactive Burden to Proactive Control
Navigating the regulatory landscape for medical devices is a constant, high-stakes challenge. Keeping technical documentation current, managing multi-jurisdictional requirements across the EU and UK, and preparing for Notified Body assessments consumes vast resources and creates significant operational risk.
RuleMedi replaces fragmented processes and manual effort with a unified, agentic AI platform that provides a single, governed source of truth for your entire compliance lifecycle — from design and certification through to post-market vigilance and continuous improvement.
One Platform. Three Integrated Solutions.
RuleMedi delivers full-lifecycle regulatory intelligence through three powerful, interconnected solutions that work in concert to ensure you are not just compliant, but in control.

Compass
The core of our platform. Compass delivers inspection-ready, evidence-backed outputs across six fully integrated pillars of regulatory intelligence — Compliance, Risk, Training, ESG, Executive Governance, and Regulatory Inspection — turning every obligation into a structured, demonstrable asset.
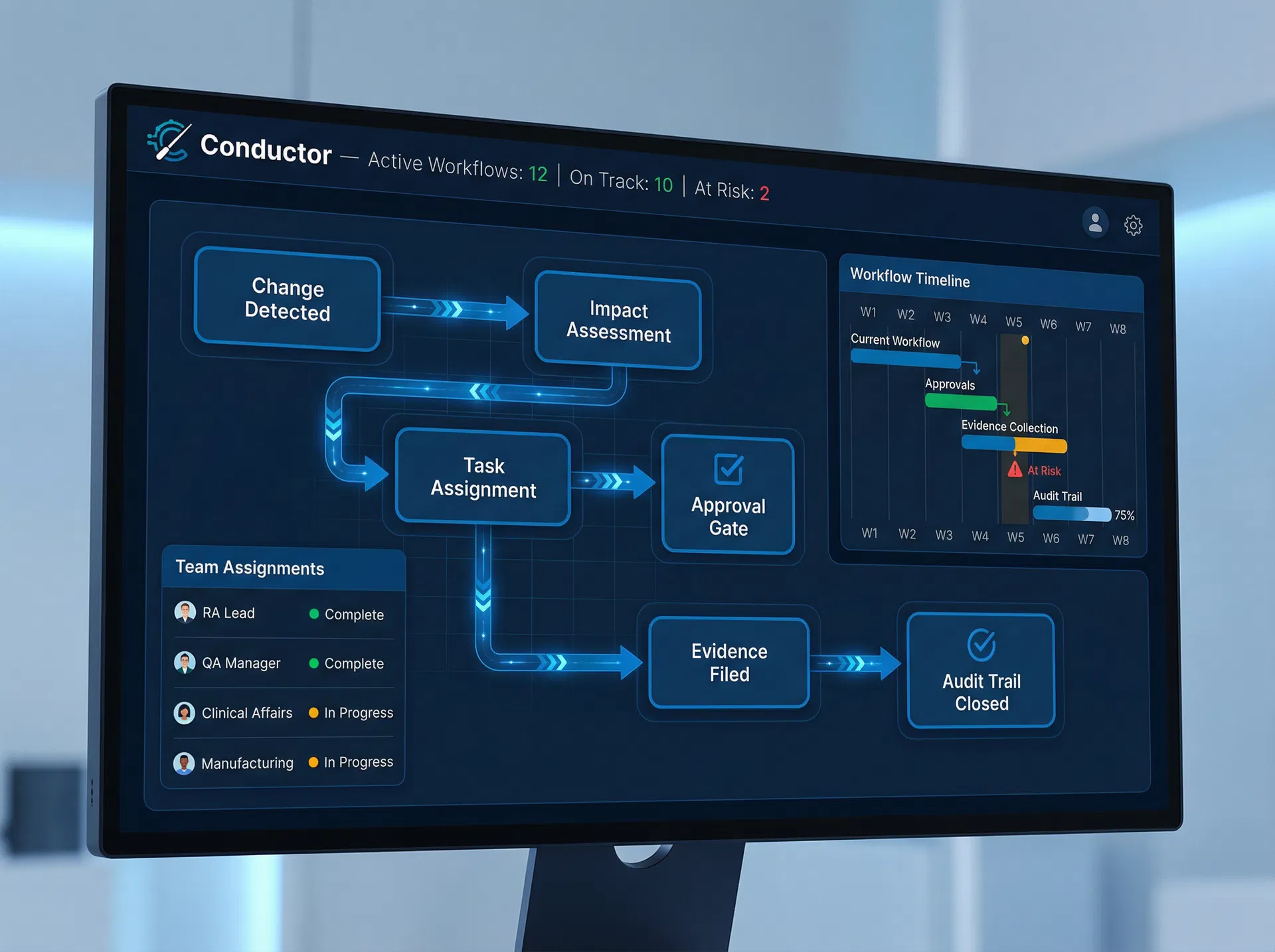
Conductor
The operational engine of RuleMedi. Conductor orchestrates people, processes, and evidence across all functions — from RA/QA and Clinical to Manufacturing and Vigilance — ensuring that compliance tasks are executed with consistent governance and a complete, unbroken audit trail.
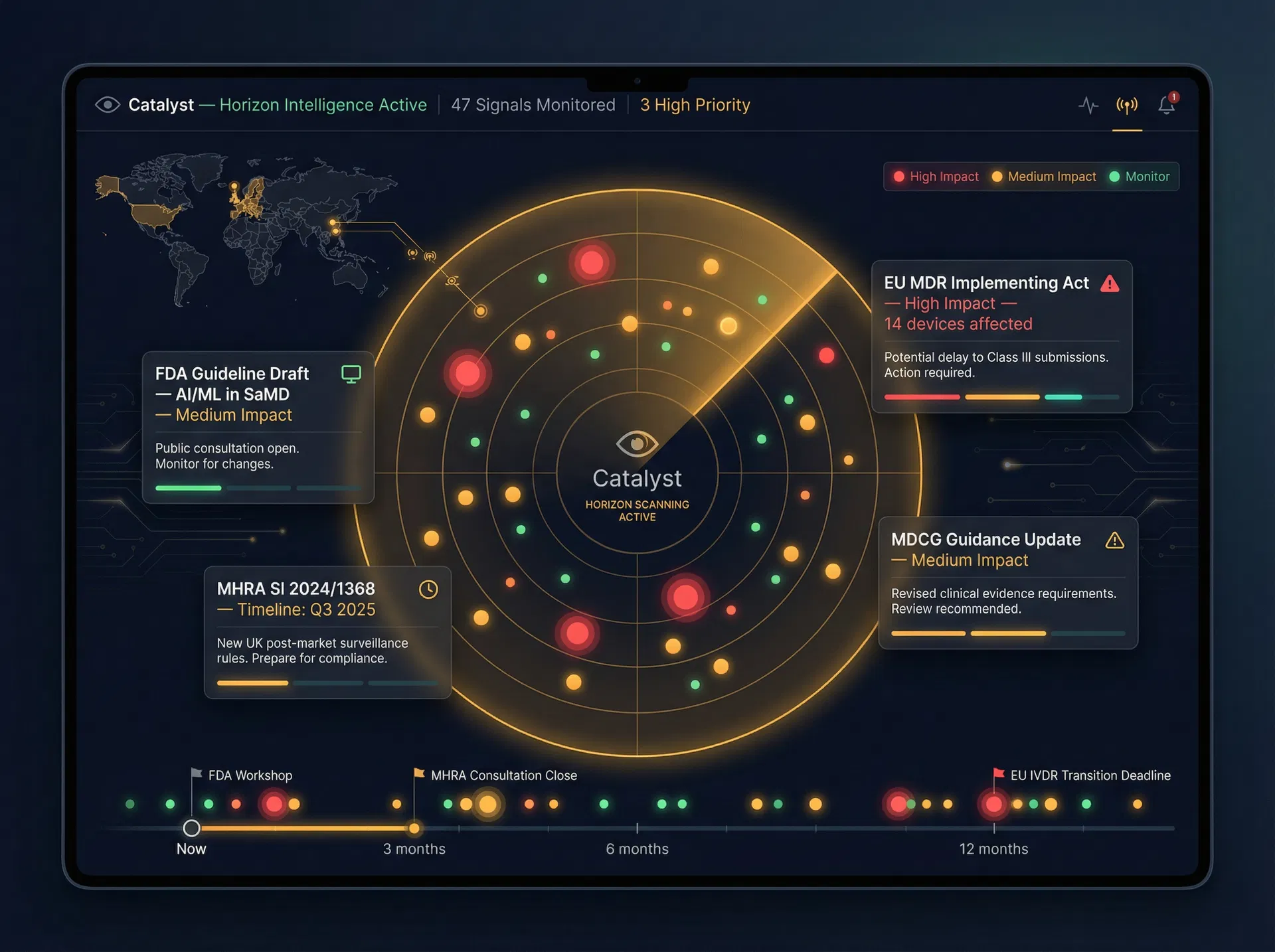
Catalyst
The strategic layer of RuleMedi. Catalyst is the anticipatory intelligence engine that scans for regulatory change, predicts its impact on your device portfolio, and provides the foresight needed to stay decisively ahead of the curve.
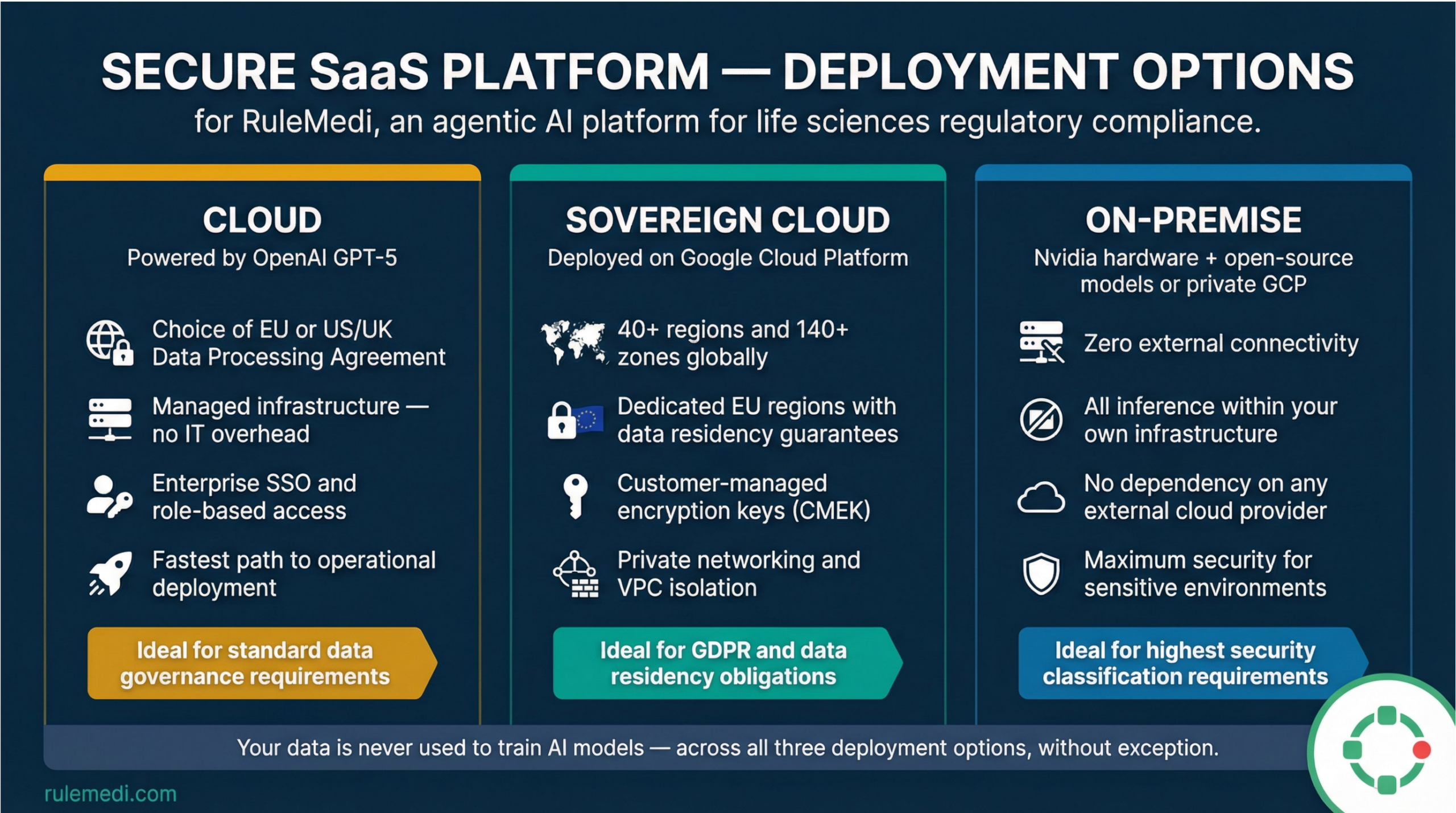
Secure, Sovereign, and Scalable Deployment
Your data security and sovereignty are paramount. RuleMedi offers three deployment options to meet the specific security, residency, and operational requirements of your organisation. Regardless of your choice, your data is never used to train AI models — and our governance standards remain absolute and unchanged.
Cloud
Powered by OpenAI GPT-5. Your organisation selects a Data Processing Agreement aligned to EU or US/UK jurisdiction. The fastest path to deployment, with fully managed infrastructure and enterprise identity controls.
Sovereign Cloud
Deployed on Google Cloud Platform across 40+ regions and 140+ zones worldwide — including dedicated EU regions. Granular, enforceable data residency with Customer-Managed Encryption Keys (CMEK) and private networking.
On-Premise
Maximum data sovereignty. AI inference runs on Nvidia hardware with open-source models, or on a private GCP environment. Optionally fully isolated, with zero external network connectivity and no dependency on any external provide – Air-Gapped.
Intelligence You Can Defend
Built by the sovereign AI experts at Duforest Ltd, RuleMedi is founded on core principles of auditable, explainable, and evidence-backed intelligence. Our platform is engineered to meet the rigorous standards of the life sciences industry and to withstand the scrutiny of Notified Bodies and regulators. Every output is version-controlled, traceable, and explainable — because in a regulated environment, credibility depends on demonstrable control.
Inspection-Ready by Design
Every output RuleMedi produces is cited, version-controlled, and traceable to source. When a Notified Body or MHRA inspector asks for evidence, your team can respond with structured, auditable documentation — not a printout from a chatbot.
Your Data. Your Control. Always.
Across all three deployment options — Cloud, Sovereign Cloud, and On-Premise — your data is never used to train AI models. You choose your infrastructure, your jurisdiction, and your encryption keys. Governance is not a feature; it is the foundation.
Built for Life Sciences. Not Adapted for It.
RuleMedi is not a general-purpose AI tool configured for compliance. It is engineered specifically for EU MDR, IVDR, UKCA, ISO 13485, and the full EU/UK regulatory ecosystem — by people who understand the difference between a technical file and a design dossier.
Built for the Full Regulatory Landscape
RuleMedi covers the complete EU, UK, and NI regulatory frameworks for medical devices and in vitro diagnostics.
EU MDR 2017/745
Medical Devices
EU IVDR 2017/746
In Vitro Diagnostics
UK MDR 2002
As Amended (UKCA)
ISO 13485
Quality Management
Ready to Transform Your Regulatory Strategy?
Move beyond compliance as a cost centre. Partner with RuleMedi to build a resilient, strategic, and continuously compliant regulatory operation that gives your leadership team the confidence to focus on innovation.