RuleMedi
Catalyst
Regulatory Foresight. Strategic Advantage. Continuous Horizon Intelligence.
Catalyst is the anticipatory intelligence engine of RuleMedi. It continuously scans the regulatory horizon for signals that will affect your device portfolio — across EU, UK, and international bodies — predicts their impact before they become obligations, and provides the strategic foresight to act decisively. In life sciences, the organisations that see change coming are the ones that shape their response. Catalyst makes that possible.
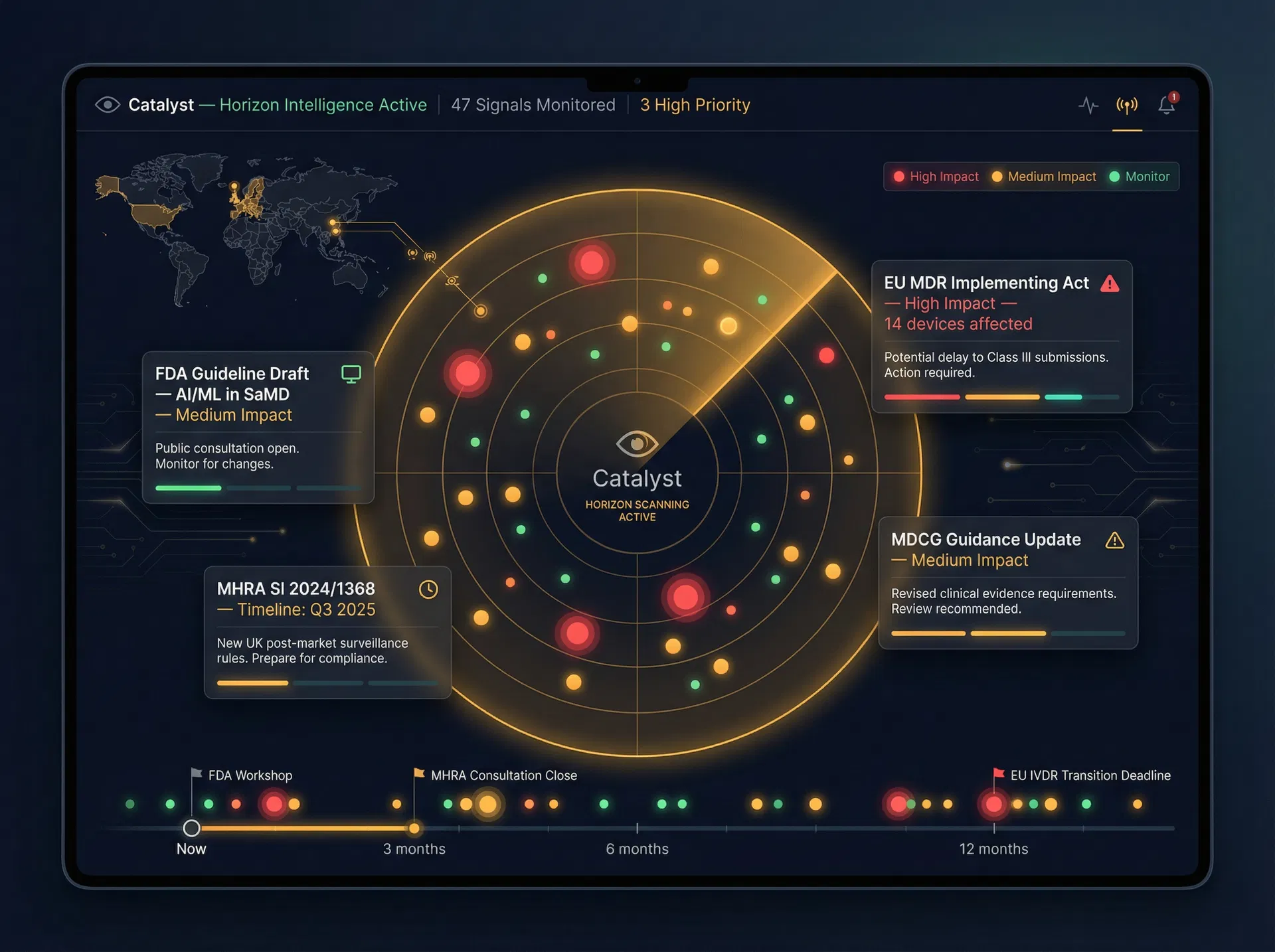
From Horizon to Action
Catalyst transforms the overwhelming volume of regulatory intelligence into a prioritised, actionable signal. It is the difference between being surprised by a regulatory change and being prepared for it months in advance.
Continuous Regulatory Horizon Scanning
Catalyst monitors regulatory pipelines across EU, UK, and international bodies — tracking proposed legislation, guidance consultations, SCENIHR and SCCS opinions, MDCG guidance, and MHRA updates — continuously, not quarterly. You are never surprised by a regulatory change.
Portfolio Impact Prediction
When a regulatory change is detected, Catalyst maps its predicted impact across your specific device portfolio — identifying which products, Technical Documentation sections, clinical evidence packages, and PMS artefacts will be affected, and by when.
Weak Signal Detection
Catalyst detects early-stage signals — enforcement trends, Notified Body audit finding patterns, SCCS opinions, and post-market safety signals — before they crystallise into formal regulatory requirements. You gain a decisive head start over competitors who wait for the official guidance.
Strategic Compliance Roadmapping
Translate horizon intelligence into actionable strategic roadmaps. Catalyst generates prioritised, time-bound compliance roadmaps that feed directly into Conductor’s workflow engine — closing the loop from intelligence to execution without manual translation.
Monitoring Every Signal That Matters
Catalyst monitors a comprehensive, structured range of regulatory intelligence sources across the EU and UK — synthesising them into a coherent, prioritised view of what is changing, what it means for your portfolio, and when you need to act.
EU Legislation
- EU MDR/IVDR implementing acts and delegated regulations.
- MDCG guidance documents and position papers
- European Commission consultation documents
UK Legislation
- SCENIHR and SCCS scientific committee opinions
- International standards updates (ISO 13485, ISO 14971, IEC 62304)
- EMA/MHRA pharmacovigilance and safety signal trend
Scientific & Technical
- SCENIHR and SCCS scientific committee opinions
- International standards updates (ISO 13485, ISO 14971, IEC 62304)
- EMA/MHRA pharmacovigilance and safety signal trends
Enforcement Intelligence
- Notified Body audit finding trends and common deficiencies
- Emerging enforcement actions and market withdrawals
- EUDAMED safety and performance data trends

Stop Reacting. Start Anticipating.
Catalyst gives your regulatory leadership the intelligence to make proactive, evidence-based decisions — turning regulatory foresight into a genuine competitive advantage for your organisation.
